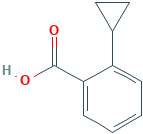
2-CYCLOPROPYLBENZOIC ACID
2-Cyclopropylbenzoic acid
CAS: 3158-74-5
Molecular Formula: C10H10O2
2-CYCLOPROPYLBENZOIC ACID - Names and Identifiers
| Name | 2-Cyclopropylbenzoic acid |
| Synonyms | AKOS BAR-1431 2-Cyclopropylbenzoic acid 2-CYCLOPROPYLBENZOIC ACID Benzoic acid, 2-cyclopropyl- |
| CAS | 3158-74-5 |
2-CYCLOPROPYLBENZOIC ACID - Physico-chemical Properties
| Molecular Formula | C10H10O2 |
| Molar Mass | 162.19 |
| Density | 1.248 |
| Melting Point | 102-106 °C |
| Boling Point | 282℃ |
| Flash Point | 131℃ |
| pKa | 4.11±0.36(Predicted) |
| Storage Condition | Room Temprature |
| Sensitive | IRRITANT |
| MDL | MFCD01550965 |
2-CYCLOPROPYLBENZOIC ACID - Risk and Safety
| Hazard Class | IRRITANT |
2-CYCLOPROPYLBENZOIC ACID - Introduction
2-Cyclopylbenzoic acid is an organic compound whose chemical formula is C10H10O2. The following is a description of the nature, use, preparation and safety information of 2-Cyclopropylbenzoic acid:
Nature:
1. Appearance: 2-Cyclopylbenzoic acid is a white to light yellow crystalline solid.
2. melting point: about 76-78 ℃.
3. Solubility: 2-Cyclopylbenzoic acid is soluble in some organic solvents, such as ethanol, ether and dichloromethane.
Use:
1. chemical raw materials: 2-Cyclopylbenzoic acid can be used as an important raw material for the synthesis of other organic compounds in the chemical industry.
2. Drug intermediates: It can be used to synthesize various drugs in the pharmaceutical field and has a wide range of application prospects.
Preparation Method:
2-Cyclopylbenzoic acid can be prepared by catalytic hydrogenation of styrene as raw material. First, cyclopropyl ethylene is prepared by controlled polymerization of the monomer of cyclopropane. Then, the cyclopropyl ethylene is subjected to cleavage to obtain 2-cyclopropyl styrene. Finally, 2-Cyclopropylbenzoic acid can be obtained by oxidative acylation of 2-cyclopropylstyrene.
Safety Information:
There is limited information on the toxicity and safety of 2-Cyclopropylbenzoic acid. Use should follow routine laboratory practices and take appropriate personal protective measures, such as gloves, goggles and protective clothing. At the same time, avoid inhalation, ingestion and contact with skin, and ensure that the place of use is well ventilated. During storage and handling, care should be taken to avoid contact with oxidants, strong acids and alkalis. If necessary, consult a chemical professional for more detailed safety information.
Nature:
1. Appearance: 2-Cyclopylbenzoic acid is a white to light yellow crystalline solid.
2. melting point: about 76-78 ℃.
3. Solubility: 2-Cyclopylbenzoic acid is soluble in some organic solvents, such as ethanol, ether and dichloromethane.
Use:
1. chemical raw materials: 2-Cyclopylbenzoic acid can be used as an important raw material for the synthesis of other organic compounds in the chemical industry.
2. Drug intermediates: It can be used to synthesize various drugs in the pharmaceutical field and has a wide range of application prospects.
Preparation Method:
2-Cyclopylbenzoic acid can be prepared by catalytic hydrogenation of styrene as raw material. First, cyclopropyl ethylene is prepared by controlled polymerization of the monomer of cyclopropane. Then, the cyclopropyl ethylene is subjected to cleavage to obtain 2-cyclopropyl styrene. Finally, 2-Cyclopropylbenzoic acid can be obtained by oxidative acylation of 2-cyclopropylstyrene.
Safety Information:
There is limited information on the toxicity and safety of 2-Cyclopropylbenzoic acid. Use should follow routine laboratory practices and take appropriate personal protective measures, such as gloves, goggles and protective clothing. At the same time, avoid inhalation, ingestion and contact with skin, and ensure that the place of use is well ventilated. During storage and handling, care should be taken to avoid contact with oxidants, strong acids and alkalis. If necessary, consult a chemical professional for more detailed safety information.
Last Update:2024-04-09 21:11:58
Supplier List
Product Name: 2-CYCLOPROPYLBENZOIC ACID Request for quotation
CAS: 3158-74-5
Tel: +86 13293139565
Email: vinnie@api-made.com
QQ: 2878563431
WhatsApp: +86 13293139565
CAS: 3158-74-5
Tel: +86 13293139565
Email: vinnie@api-made.com
QQ: 2878563431
WhatsApp: +86 13293139565
Product Name: 2-CYCLOPROPYLBENZOIC ACID Request for quotation
CAS: 3158-74-5
Tel: +86 13293139565
Email: vinnie@api-made.com
QQ: 2878563431
WhatsApp: +86 13293139565
CAS: 3158-74-5
Tel: +86 13293139565
Email: vinnie@api-made.com
QQ: 2878563431
WhatsApp: +86 13293139565
View History
2-CYCLOPROPYLBENZOIC ACID
3,9-Diazaspiro[5.5]undecane-3-carboxylic acid, 1,1-dimethylethyl ester, hydrochloride (1
3,9-Diazaspiro[5.5]undecane-3-carboxylic acid, 1,1-dimethylethyl ester, hydrochloride (1
